Introduction
Epilepsy is an important neurologic manifestation of patients with tuberous sclerosis complex (TSC). The reported prevalence of epilepsy in TSC is about 62% to 93%.1–3 Epilepsies associated with TSC usually present during infancy and early childhood, but it can also occur at any age. The most common type of seizures are focal seizure and epileptic spasm. However, the pattern of seizures in TSC changes over time. About half of the patients have refractory epilepsy.4–9 There are limited numbers of studies performed on TSC in developing countries. Thus, this study was done in children with TSC with epilepsy to evaluate the types of seizure, electroencephalography (EEG) features, neuroimaging pattern, drug treatment and response to treatment.
Methods
Study protocol
This study was carried out at a tertiary care center in Bangladesh. It was conducted on patients with TSC and epilepsy, children of all age (0–18 years), were included during the period of January 2011 to October 2019. Patients were diagnosed as a case of TSC according to TSC clinical diagnostic criteria.10 initially, 84 patients were included, the data of seventy patients were ultimately used as adequate data for fourteen patients were missing. It was an observational study.
Detail history regarding seizure semiology, family history, developmental status, treatment history were taken. Formal clinical examination with special emphasis on eye, skin, abdomen, chest, nervous system, developmental assessment and psychological evaluation were performed. Neurobehavioural disorders like autism spectrum disorder and attention deficit hyperactivity disorder were diagnosed with Diagnostic and Statistical Manual V (DSM V). Intellectual disability was diagnosed when IQ was less than 70. The tools used here were Bayley Scales of Infant Development III, Wechsler Preschool and Primary Scale of Intelligence III, and Wechsler Intelligence Scale for Children-Revised IV.
For all patients a neuroimaging, preferably magnetic resonance imaging (MRI) of brain, and EEG were conducted. For target organ screening, color Doppler echo, USG of whole abdomen, X-ray chest, formal ophthalmological evaluation were performed. Patients were followed up 6-monthly for a period of at least 1 year.
Results
Patient demographics
In this study seventy patients were analyzed. Mean age of the patients was 5.64±3.96 years. Among them 62 patients (88.6%) were bellow or equal to 10 years old. There was slight female predominance (57.1%) among the patients. Only 7.1% of patients had positive family history of TSC (Table 1).
Clinical manifestations of TSC
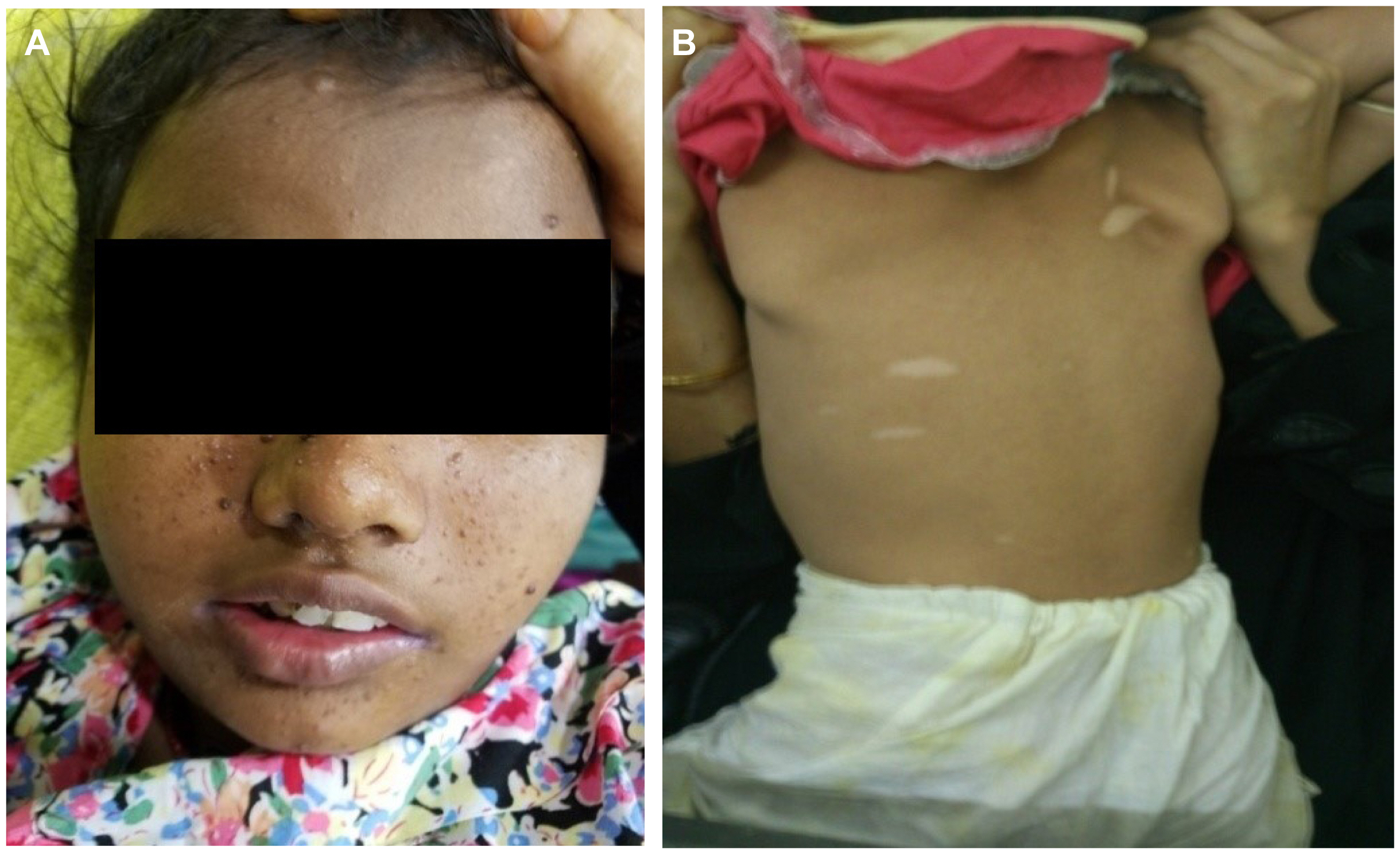
Regarding skin manifestation, all the children had skin manifestations; most common finding was ash leaf spot (74%). Other skin findings were adenoma sebaceum (60%), shagreen patch (33%), café au-lait spot (4.28%), and subungual fibroma (Fig. 1). Most of the patients had multiple types of skin findings. Apart from neurological findings and skin manifestations, other systems involvement was minimal, namely four patients with cardiac involvement (rhabdomyoma and tricuspid valve regurgitation), two patients with renal involvement (angiomyolipoma and renal cyst), one patient with oral fibroma, and two patients with cataract. No patient was found with chest finding (Table 2).
Seizure in studied patients (Table 3)
Onset of seizure
Thirty-seven percent of the patients had the onset of seizure in the first 6 months of life. About 26 percent (25.7%) of patients had the onset of seizure after 5 years of age. Thus, in most of the patients, seizure onset was in first year of life (60%) (Table 3).
Type of seizure
The most prevalent type of seizure was focal seizure occurring in about 46% of patients. Epileptic spasm occurred in 17% of patients and generalized seizure in 27% of patients. However, 4% of patients had a history of epileptic spasm followed by focal seizure and 5.7% of patients had other types of seizure. All the patients who had epileptic spasm presented in less than 1 year of age (Table 3).
Developmental status of the studied children
Regarding developmental status, more than half of the children (54.3%) had developmental delay in at least one domain and thirty children (42.9%) had normal development. While two patients (2.9%) had regression of development (mainly cognition and motor), both of them had intractable epilepsy (Table 1).
Electroencephalographic findings
All the patients with epilepsy had abnormal EEG. Focal epileptic discharge was the most prevalent EEG change in this study group (47%). Hypsarrhythmia was found in ten patients (two classical and eight modified hypsarrhythmia). While 17 patients (24%) had generalized discharge, eight patients (11.4%) had epileptic encephalopathy and two patients had the EEG suggestive of Lennox-Gastaut syndrome (Table 3).
Neuroimaging findings
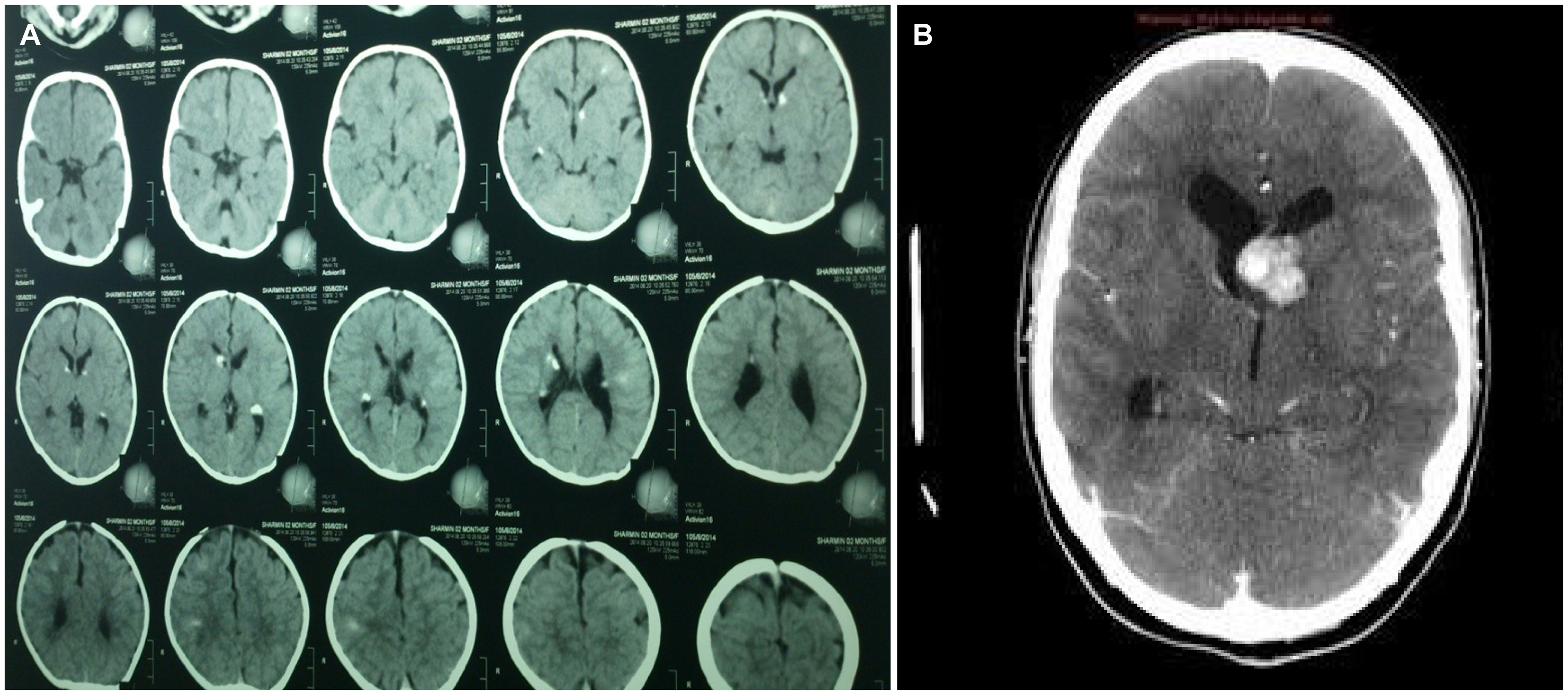
Neuroimaging (computed tomography scan or MRI of brain) were performed in all the studied subjects. 50% (35) of patients had both subependymal nodules (SEN) and cortical tubers, while 24% (17) had only SEN and 21% (15) had only cortical tubers. One patient had subependymal giant cell astrocytoma (SEGA) along with SEN. Two other patients had multiple central nervous system findings; one patient had both SEN and temporal cyst while another patient had SEN, cortical tuber and cyst. No patient had white matter abnormality or hydrocephalus (Table 4, Fig. 2).
Antiepileptic drugs (AEDs) used
Regarding AED use, more than half of the patients (55.7%) received vigabatrin either as a single agent or in combination with other drugs. Among these patients taking vigabatrin, 16 patients had epileptic spasm (ES). In this cases, vigabatrin was started as the first or second line drug. Eight patients with IS completely responded to vigabatrin while three patients had partial response and in one patient vigabatrin was withdrawn due to somnolence. In this patients injection adrenocorticotropic hormone (ACTH) was given. Twenty six percent of children in this study group got two AED, 20% got three drugs, 16 % got four AED and 4% got five AED. In our cohort, 34.2% of patients were seizure free for 12 months, 22.8% had drug resistant epilepsy, 38.5% were seizure free for at least 3 months with 1 or 2 drugs, and 4.2% had relapse (Table 5).
Treatment with mammalian target of rapamycin (mTOR) antagonist
Fifteen patients (21.4%) of this study group got mTOR antagonist (everolimus) for a period of 2 months to 1 year. Out of this, six patients were seizure free, but it was an add-on with other AEDs. Out of 15 patients, one patient had SEGA which showed decrement of size after treatment.
Short term outcome of the studied children
In follow-up, 24 patients (34.2%) were seizure free for at least 1 year, 16 patients (22.8%) had ongoing seizure and 27 patients (38.5%) had epilepsy controlled for at least 3 months with drug. Three patients had epilepsy relapse (Table 3).
Discussion
This study is a longitudinal observational study of children with TSC with epilepsy in a developing country like Bangladesh. Here all the patients were managed at a tertiary care centre. This study will contribute in better understanding of epilepsy and other co-morbidities in children with TSC along with the pattern of EEG and unique neuroimaging findings. This study has been performed in 70 patients who have TSC and epilepsy with mean age of 55.64±3.96 years.
Although epilepsy is not diagnostic criteria of TSC, it is an important neurological feature. The prevalence of epilepsy in TSC is as high as 90%.1,11 In this study group the most prevalent form of seizure was focal seizure. In about 46% of children, focal seizure was the initial pattern of seizure while in 4.3% of patients, focal seizure occurred after epileptic spasm. This finding coincides with the study done by Devis et al.,12 who found focal seizure in 55% of patients. They found both focal seizure and IS in about 36% of patients, which is quite higher from our study finding.12 In a TuberOus SClerosis registry to increase disease Awareness (TOSCA) study also, focal seizure was the most prevalent type of seizure occurring in 67.5% of patients.13
Generalized seizure was the second most common type of seizure in this study group occurring in about 27% of children. Among them, 13 (68%) had focal onset. Remaining six children (32%) had true primary generalized epilepsy. A similar study done by Chu-Shore et al.1 revealed that about 37% of patients had generalized seizure although most of them had focal onset like our study. They also suggested that the number of patients with true generalized epilepsy in TSC may be overestimated as here the categorization has been performed based upon the clinical description, not the ictal EEG recording. As TSC neuropathogenesis favors the focal onset of the seizure, it is most likely that most of the seizures are partial in onset with or without secondary generalization.1
In this cohort, 17% of patients had epileptic spasm and this is a bit lower than the prevalence of epileptic spasm in a TOSCA study and a study done by Chu-Shore et al.,1 where the overall prevalence is about 38.9% and 37%, respectively.13 The occurrence of epileptic spasm is higher in early ages, particularly in the first 2 years which is about 46% of patients in some studies.14
Usually in TSC, the onset of seizure is in early childhood, mostly in infancy, although they can occur at any age. Moreover, patients remain at risk of epilepsy, particularly focal epilepsy, throughout their lifetime.12 In our cohort, 37% of patients had their first seizure before 6 months and 23% had it between 6 to 12 months. Thus, the onset of seizure bellow 1 year was 60%. However, delayed onset seizure was not uncommon. About 25.7% of patients had their first seizure after 5 years of age. Our study result thus matches with that of TOSCA study, where 79% of patients had their first seizure before 2 years. They also noticed the late onset of seizure even at the age of 40 years. In this study group, they have noticed that occurrence of IS was higher in less than 2 years, but the occurrence rate of focal seizure was equal in both early and late onset seizure.13
EEG plays an important role for diagnosis, prognosis and treatment of epilepsy in TSC.13 In our cohort, all the patients had abnormal EEG. Most common type of abnormality found are focal discharges arising from temporal and occipital areas. Some of them were multifocal in origin. In cases of epileptic spasm, 14.3% had hypsarrhythmia and the rest of them have multifocal discharges.
The commonest neuroimaging finding was subependymal nodule (78.9%), while other findings were cortical tuber in 21.4% of patients, SEGA in one patient, and arachnoid cyst in one patient. In a related study done by Davis et al.,12 they found SEN in 90% of patient, both SEN and cortical tuber in 89% of patients, and normal neuroimaging in 4% of patient. In our cohort, we did not find any normal neuroimaging. It is here to mention that; neuroimgaing in TSC may initially appear normal and might found in follow up study.12
Meticulous choice of AED is very important in TSC. Seizure control in an early stage is closely related to better psychomotor outcome.1,14 In our cohort, 34.2% of patients went to remission, 22.8% had drug resistant epilepsy, 38.5% had epilepsy with 1 or 2 drugs, and 4.2% had relapse. Treatment with GABAergic drugs (i.e., vigabatrin) plays a vital role in TSC. The current recommendation is vigabatrin, which should be used as a first line AED treatment for epileptic spasm with TSC and for focal seizure before the age of 1 year.4,15 Almost all the patients with epileptic spasm received vigabatrin in our cohort, but as one patient developed Lennox-Gastaut syndrome, we did not gave him vigabatrin. Other drugs used here were Na valproate, levetiracetum, phenobarbitone, benzodiazepine, oxcarbazepine, carbamazepine, topiramate, and oral steroid etc. In one patient who had epileptic spasm and developed side effect of vigabatrin, injection ACTH was given. Seizure was controlled with the administration of 1 or 2 drugs in most of the patients; very few patients needed more than 2 drugs. Drug resistant cases were 22.8%, which has similarity with that of TOSCA study were, in epileptic spasm the resistant cases were 15.5%, and in focal seizure it was 38%.16
The recent Examining Everolimus in a Study of Tuberous Sclerosis Complex (EXIST) 3 study shows higher efficacy of adjuvant everolimus therapy in patients with drug resistant seizure associated with TSC compared to placebo.17 Seizure relapse is not uncommon in TSC. In a study by Sparagana et al.,18 more than one forth patients experiences relapse while Chu-Shore et al.1 found 9.1% of patients had epilepsy relapse. In our study it was much less, about 4.2%. This discrepancy may be due to short time follow up and the difference of the population studied.1 However, it is importnant to taper AEDs in patients when the seizure is well controlled and improved or resolved EEG abnormalities. Although there are different studies regarding the ketogenic diet, surgery and vagus nerve stimulation in TSC with epilepsy, none of the modalities have been used in this study.17,19
Apart from epilepsy and neurological stigma, there is the involvement of multiple organs in TSC. The commonest cutaneous finding in this cohort was ash leaf spot (74%). We also searched for target organ involvement and found that heart was most commonly affected in our cohort. Along with this, renal, eye, and oral involvement was detected in this cohort.
This prospective study demonstrates the pattern of seizure, EEG changes, neuroimaging findings of children with TSC with epilepsy. It also highlights the response to therapy. In early infancy common forms of seizure were epileptic spasm and focal seizure. It also confirms that epilepsy with TSC is difficult to treat as only 34.2% of patients had a sustained seizure freedom and 4.2% had relapse. Also, a good number of patients needed more than two drugs to control seizure.