Introduction
Benign childhood epilepsy with centrotemporal spikes (BCECTS) is the most common focal epilepsy syndrome in children, accounting for 10–24% of pediatric epilepsies. Age at BCECTS onset varies greatly, from 2 to 13 years of age, and peaks at approximately age 7–10.1 Patients experience partial seizures with orofacial involvement and frequent involvement of one or both hands, as well as of the leg ipsilateral to the affected side of the face. Patients occasionally experience generalized seizures.2,3 Seizures typically occur during non-rapid eye movement sleep.1 Electroencephalography (EEG) shows high-voltage spikes or spike-and-wave complexes in the centrotemporal region that may spread to the contralateral side.2
The prognosis is typically excellent, with remission of seizures often occurring 2–4 years after initial onset and usually before age 16.4 However, in 1–7% of patients with BCECTS, the condition may evolve into more severe syndromes such as atypical benign childhood focal epilepsy, Landau-Kleffner syndrome, or occurrence of continuous spikes and waves during slow wave sleep.5 Most patients with BCECTS experience seizures rarely, and therefore, do not require treatment with antiepileptic drugs (AEDs). Patients with recurrent seizures typically respond well to AED treatment. However, some patients do not respond well to the initial drug and may therefore require more than 1 AED to control seizures.
Few previous studies have assessed prognostic factors associated with poor initial responses to AEDs. Therefore, we conducted a retrospective study to compare two groups of children with BCECTS, one group using 1 AED and the other group using more than 1 AED, in order to identify risk factors associated with poor responses to initial AED therapy.
Methods
Patients and methods
We retrospectively reviewed the medical files of 145 patients diagnosed with BCECTS at Korea University Medical Center between January 2008 and September 2013. Participants met all criteria for diagnosis of BCECTS, as outlined by the International League Against Epilepsy in 1989. Patients had brief focal or generalized seizures during sleep with the centrotemporal spikes in EEG. And onset occurred between the ages of 3 and 13 years. Patients with atypical benign partial epilepsy or obvious neurological deficits were excluded from the study. The minimum follow-up period was 1 year.
Out of 145 patients, 88 (61%) were not treated with AEDs, 41 (28%) were treated with 1 AED, and 16 (11%) were treated with 2 AEDs. Because we sought to identify risk factors associated with poor response to initial AED therapy, untreated patients were excluded. We placed the remaining patients into two groups: patients being treated with only 1 AED (good responses), and patients being treated with 2 AEDs (poor responses). Data retrieved from medical records included age of the patient, gender, age of afebrile seizure onset, number of afebrile seizures experienced before beginning drug therapy, history of febrile seizures, family history of febrile seizures or epilepsy, brain magnetic resonance imaging (MRI) and EEG findings, type of seizures, and use of AEDs.
EEGs were recorded with 18 electrodes during wakefulness and sleep. Localization of spikes and waves to unilateral or bilateral centrotemporal areas was noted. The presence of generalized epileptiform discharges or focal slowing was also recorded.
This study was approved by the Institutional Review Boards of Korea University’s Anam, Guro, and Ansan Hospitals.
Statistical analyses
Data were analyzed using SPSS software (version 20, SPSS Inc., Chicago, IL, USA). Chi-square tests were used to identify differences between groups. Multiple logistic regression with binary dependent variables was conducted to identify prognostic factors for poor response to initial AED therapy. The forward likelihood ratio method was used in variable selection. Statistical significance was defined as p <0.05.
Results
Demographic and clinical data
Our retrospective study included 57 patients with BCECTS, including 30 male (53%) and 27 female (47%) children. The mean age of afebrile seizure onset was 7.3 ± 2.5 years (range: 3–13 years). Of these patients, 14 (25%) had unilateral motor seizures, and 43 (75%) had generalized seizures. Before beginning drug therapy, 23 patients (40%) had experienced fewer than three seizures, and 34 (60%) had experienced three or more seizures. 11 patients (19%) had a past history of febrile seizures, 7 (12%) had a family history of febrile seizures, and 6 (11%) had a family history of epilepsy. 7 patients (12%) showed atypical brain MRI findings. 2 of these patients had suspected hippocampal sclerosis, 1 had mild dilatation of the right lateral ventricle without an obstructing lesion, 1 had choroid cysts, 1 had an enlarged cavum vergae, 1 had temporal lobe asymmetry without focal lesions, and 1 had suspected hypoplasia of the anterior falx cerebri.
EEG recordings were performed in all patients in both awake and sleep states. EEG findings indicated that 12 patients (21%) had generalized epileptiform discharges, and 2 patients (4%) had focal slowing. Unilateral centrotemporal spikes occurred in 22 patients (39%), and 35 patients (61%) showed bilateral centrotemporal spikes.
Response to AED treatment
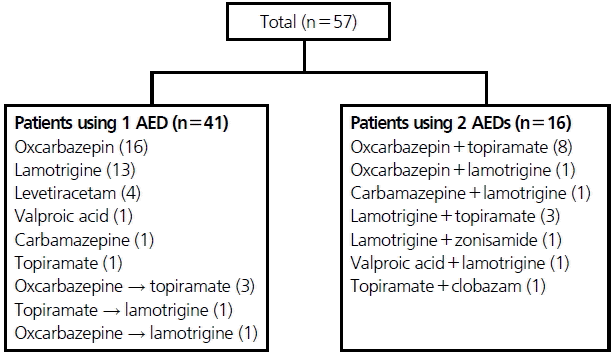
Patients were categorized into two groups. The first group included 41 patients (72%) who were successfully treated with only 1 AED (good responses). The second group included 16 patients (28%) who did not respond well to initial AED treatment and were therefore treated with 2 AEDs (poor responses).
The mean follow-up period for patients treated with AEDs was 3.17 ± 1.35 years (range: 1.03–5.89) (Table 1). Initial AEDs used included oxcarbazepine (51%), lamotrigine (30%), levetiracetam (7%), topiramate (5%), carbamazepine (4%), and valproate (4%). The second AEDs added to the treatment regimen, when applicable, included topiramate (69%), lamotrigine (19%), zonisamide (6%), and clobazam (6%) (Fig. 1).
Risk factors associated with poor response to initial AED treatment
According to a multiple logistic regression analysis, initial AED response was significantly associated with early onset of afebrile seizures (age ≤ 5 years) and with history of febrile seizures. However, no significant associations were found between initial AED response and gender, type of seizures, number of afebrile seizures experienced before beginning drug treatment, family history of febrile seizures or epilepsy, brain MRI findings, lateralization of EEG results, or focal slowing or generalized epileptiform discharges recorded by EEG (Table 2).
In conclusion, early onset of afebrile seizure (age ≤ 5) and history of febrile seizures were risk factors for poor response to initial AED therapy.
Discussion
Recurrent seizures associated with BCECTS are typically well controlled with AEDs, and spontaneous remission occurs for most patients.6 Approximately 10–13% of patients with BCECTS experience only 1 seizure. However, approximately 20% of patients experience intractable seizures, which may occur several times each day. For most patients, seizure remission occurs by 15–16 years of age.7 A meta-analysis study indicated that remission occurs in 50% of patients by 6 years of age, 92% by 12 years of age, and 99.8% by 18 years of age.8 However, some patients experience a longer active seizure period and frequent seizure recurrence. The purpose of the present study was to determine the risk factors associated with poor response to initial AED treatment in patients with recurrent seizures.
We found that early onset of afebrile seizures (age ≤ 5) was a risk factor for poor response to initial AED therapy, consistent with results of previous studies. Kramer et al. demonstrated that seizure onset prior to 3 years of age predicts the occurrence of multiple seizures.3 You et al. reported that early seizure onset (age<3) is associated with more frequent seizures that are refractory to initial medical treatment.9 Additionally, Incecik et al. demonstrated that early age of seizure onset (age<4) is an independent risk factor predicting poor response to initial AED treatment.10
Few studies have investigated the relationship between BCECTS and history of febrile seizures. You et al. and Incecik et al. found that there was no correlation between response to initial AED treatment and history of febrile seizures.9,10 In contrast, the results of our study reveal that history of febrile seizures is a risk factor for poor response to initial AED treatment, as indicated by multiple logistic regression analysis. Kajitani et el.11 suggested the possibility of a genetic link between BCECTS and febrile seizures. Their study compared the previous and family histories of febrile convulsion of 100 children with BCECTS (ages 3–13 years) with those of 100 non-epileptic control participants matched for age and sex. Children with BCECTS showed a significantly greater incidence of febrile seizures compared to non-epileptic controls (18% and 8%, respectively; p <0.05).
Carbamazepine, lamotrigine, levetiracetam, oxcarbazepine, and valproate are AEDs recommended for initial treatment by the United Kingdom National Institute of Health and Care Excellence.12 However, there are considerable geographic differences in the prescription of commonly used drugs. Carbamazepine and oxcarbazepine are most commonly prescribed in the USA,13 whereas valproate is most commonly used in Europe.14 Sulthiame is typically prescribed in Germany, Austria, and Israel. In the present study, oxcarbazepine (51%) and lamotrigine (30%) were the most commonly prescribed AEDs. Several previous studies have assessed the efficacies of various AEDs. One study reported that 16 of 18 patients (89%) treated with clobazam did not experience seizures during the 9 months of the study period, compared to 21 of 25 patients (84%) treated with carbamazepine.15 Another study found that 19 of 21 patients (91%) using levetiracetam and 13 of 18 patients (72%) using oxcarbazepine experienced seizure remission.16 Borggraefe et al.13 reported seizure recurrence in 4 patients (19%) using levetiracetam and in 2 patients (9%) using sulthiame. Overall, however, there is insufficient evidence regarding the appropriate choice of AEDs. Therefore, physicians should exercise caution when deciding to treat patients with AEDs and consider patient characteristics when choosing a specific AED.
This study did not consider differences in drug efficacies, which may influence patient prognosis. Furthermore, this is a retrospective study, which is a significant limitation. Further studies should use different study designs in order to identify additional risk factors for poor response to initial drug therapy in patients with BCECTS.
BCECTS is the most prevalent focal epilepsy syndrome in children and is considered benign and easy to control with AEDs. Although seizures in some patients are refractory to initial medical treatment, the long-term prognosis is good. In this study, we have identified early onset of seizures (≤ age 5) and history of febrile seizures as predictors of poor response to initial treatment.