Introduction
Recently, many new antiepileptic drugs (AEDs) have been developed, which has led to additional options for the care of patients with epilepsy. New AEDS are needed because there are still patients with epilepsy that is refractory to established AEDs. Furthermore, there is a need for new AEDs without pharmacokinetic drug interactions, drugs with different mechanism of action to allow synergistic combination therapy, and drugs with fewer adverse events (including idiosyncratic, teratogenic, and cognitive ones) compared with old AEDs. Therefore, it has become more important to gain understanding of the efficacy, mechanisms of action, and adverse-event profiles of new AEDs and drug interactions between these drugs, which play a key role in rational and effective combination therapy. However, the efficacy and role of new AEDs have not been elucidated fully.
The effect of initial monotherapy
Achieving complete seizure remission without adverse events is the goal of epilepsy treatment. The National Institute for Health and Care Excellence (NICE) guidelines also point out that the aim of treatment is to abolish seizures completely, while also keeping the side effects of treatment to a minimum so that patients can lead a life that is as normal as possible.1
It is well known that more than 30% of patients continue to have seizures despite pharmacological treatment. In other words, up to 70% of patients’ seizures can be well controlled by AEDs,2 although some patients experience fluctuating courses between remission and seizure recurrence.3,4 The probability of seizure freedom in patients with newly diagnosed epilepsy declines rapidly after the failure of the first monotherapy. Most patients with well-controlled epilepsy respond well to the initial AED. Therefore, the selection of the initial AED depends on the characteristics and frequency of AED adverse events to a greater extent. If efficacy is not very different between AEDs, the propensity and nature of adverse events are the determining factors in the choice of AED.
Although the good efficacy of the initial monotherapy is well known, the relative efficacy of new AEDs over old AEDs has not been established. New AED approvals are achieved by add-on trials of patients with refractory epilepsy. A 50% responder rate is the usual threshold for the approval of new AEDs. This system cannot represent all patients in real clinical practice, especially patients who need initial monotherapy.5
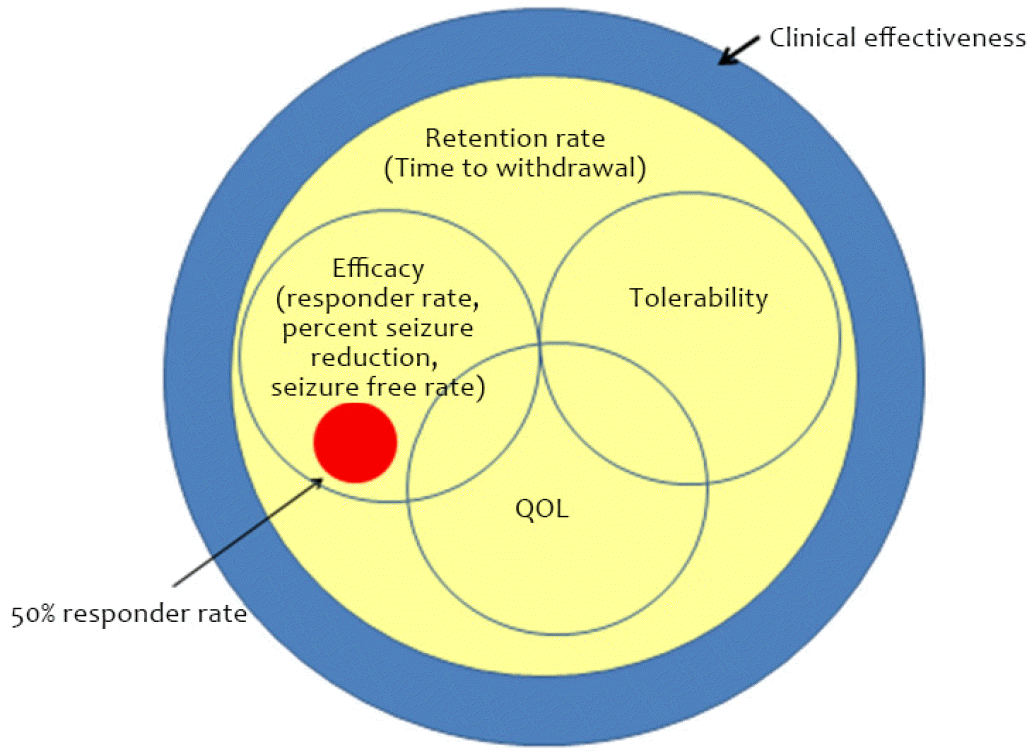
Only a few studies have been conducted to evaluate this issue. The SANAD trial was designed in an unblinded, randomized fashion to evaluate the effectiveness of carbamazepine, gabapentin, lamotrigine, oxcarbazepine, or topiramate in the treatment of partial epilepsy.6 The comparison parameters were time to treatment failure, which was defined as the discontinuation of randomized drugs because of either inadequate seizure control or intolerable side effects. Another parameter was time to 1-year seizure remission. Time to treatment failure represented the retention rate. The retention rate encompassed the efficacy, tolerability, and quality of life (Fig. 1). In summary, regarding time to treatment failure, lamotrigine was significantly better than carbamazepine, gabapentin, and topiramate, and exhibited a nonsignificant advantage compared with oxcarbazepine. Regarding time to 1-year remission, carbamazepine was significantly better than gabapentin, but exhibited a nonsignificant advantage against lamotrigine, topiramate, and oxcarbazepine. In other words, even though some new AEDs yielded a better retention rate, they failed to show better efficacy than old AEDs.
In another SANAD trial, the effectiveness of valproate, lamotrigine, or topiramate to treat generalized and unclassified epilepsy was evaluated.7 Regarding time to treatment failure, valproate was significantly better than topiramate, but there was no significant difference between valproate and lamotrigine. Valproate was significantly better than lamotrigine in time to 1-year remission, but there was no significant difference between valproate and topiramate. Therefore, for the treatment of generalized or unclassified epilepsy, new AEDs also failed to show better efficacy than old AEDs.
One randomized double-blinded trial was performed to compare the efficacy of levetiracetam and controlled-release carbamazepine (carbamazepine CR) in patients with newly diagnosed epilepsy.8 The 6-month seizure-freedom rate was almost the same for both drugs: 73.1% for levetiracetam and 72.8% for carbamazepine. Time to withdrawal was also almost identical. Fewer patients on levetiracetam (14.4%) discontinued treatment because of adverse events compared with carbamazepine CR (19.2%), although this difference did not reach statistical significance.
Recently, the KOMET study was completed to compare the efficacy of carbamazepine CR and extended-release sodium valproate (val-proate ER), as well as levetiracetam in patients with newly diagnosed epilepsy.9 This was a two-parallel-group, stratified trial. If the physician thought that valproate ER was the best treatment, the patients were randomized into treatment with valproate ER or levetiracetam. When the physician regarded carbamazepine CR as the best choice, the patients in this stratum were randomized into levetiracetam or car-bamazepine CR arms. The conclusion of this trial was simple: levetiracetam was not superior to either valproate ER or carbamazepine CR regarding the time to treatment withdrawal or time to first seizure.
If the efficacy of new AEDs is not superior to that of old AEDs, what is the meaning of developing and using new AEDs?
Combination therapy
More than 30% of patients do not respond well to AEDs. Patients with symptomatic or cryptogenic epilepsy are more likely to exhibit continuation of seizures.10 Combination therapy or epilepsy surgery can be considered for these patients. When combining AEDs, many important factors should be considered, including efficacy, adverse events, spectrum, pharmacokinetic drug interactions, pharmacodynamic interactions, titration speed, and speed of action onset of AEDs.
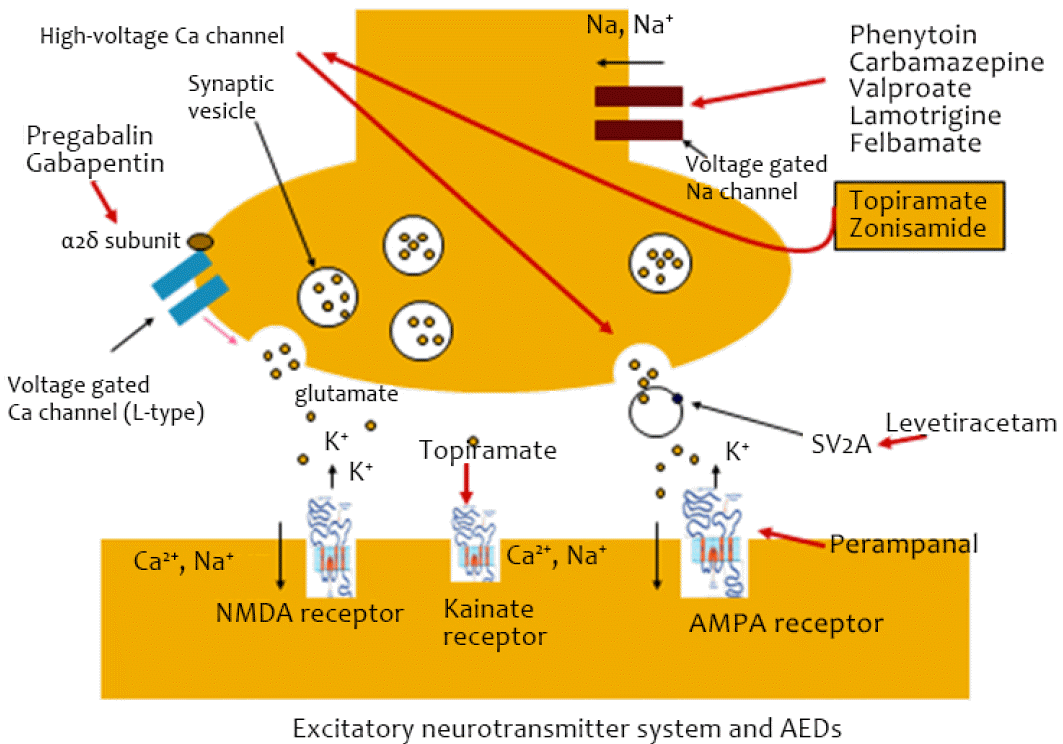
Pharmacodynamics is the mechanism of action of AEDs and the manner in which AEDs exert their effect on the target organ for the generation of programmed action. AEDs can exert their antiepileptic effect via the inhibition of excitatory neurotransmitter systems or the enhancement of inhibitory neurotransmitter systems. There are many different mechanisms of action for various AEDs.11 Moreover, some AEDs have multiple mechanisms. In excitatory neurotransmitter systems, phenytoin, carbamazepine, valproate, lamotrigine, felbamate, topiramate, and zonisamide involve voltage-gated sodium channels (Fig. 2). Lacosamide acts on voltage-gated sodium channels and is unique in the slow inactivation of sodium channels. Topiramate and zonisamide also have an effect on high-voltage calcium channels. Pregabalin and gabapentin bind to the α2δ subunit of voltage-gated calcium channels. Levetiracetam has a unique mechanism, as it binds to the SV2A protein of synaptic vesicles, which functions in synaptic modulation. A few drugs have a direct effect on receptors. Topiramate and perampanel may have an effect on kainate receptors and on the AMPA receptor, respectively.
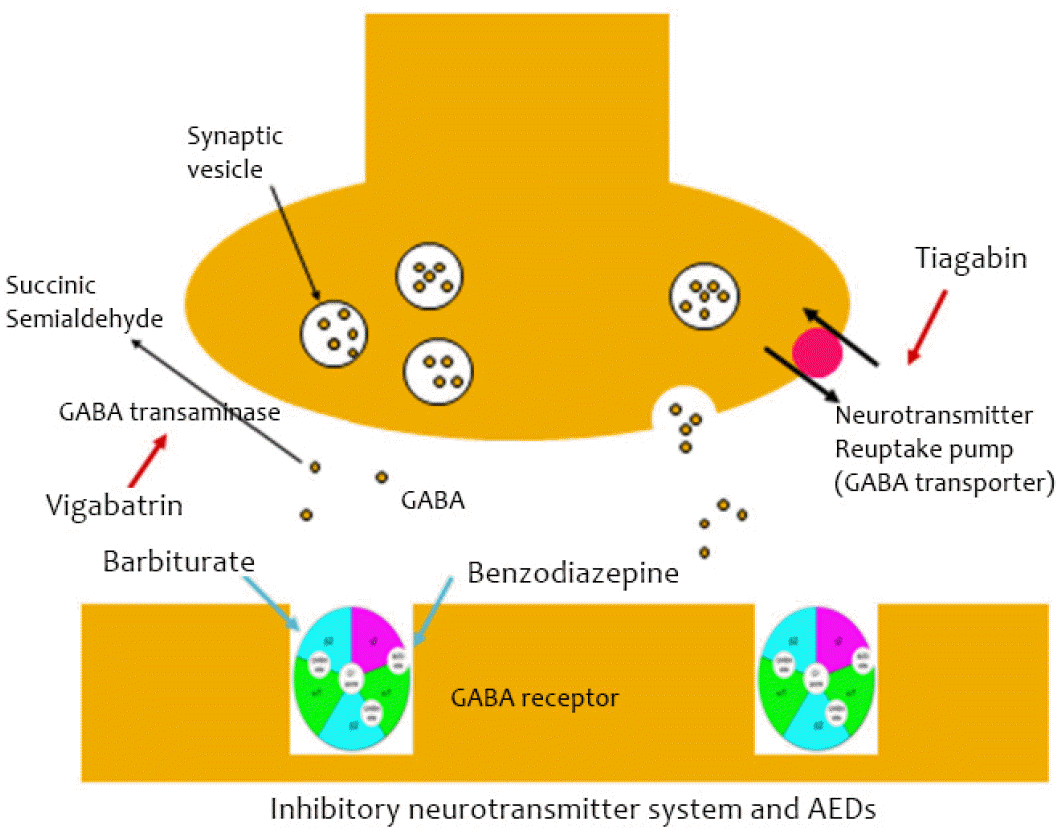
In inhibitory neurotransmitter systems (Fig. 3), barbiturate and benzodiazepine bind directly to the GABA receptor, which hyperpolarizes cell membranes by opening chloride channels. Tiagabin inhibits GABA reuptake in the presynaptic membrane, which leads to the accumulation of GABA in the synaptic cleft. The same effect can be achieved by vigabatrin, by binding irreversibly to GABA transaminase (which metabolizes GABA).
When combining drugs, in theory, the combination of AEDs with different mechanisms may have a higher chance of synergistic effects.12 A synergistic effect implies that the effect of the combination of two drugs is greater than just the simple sum of the effects of the individual drugs.13 An animal experiment showed that some drug combinations might exhibit synergism.12 For example, oxcarbazepine plus levetiracetam or lamotrigine plus valproate may have advantages in this sense. Some combinations of AEDs sometimes exacerbate adverse events. The combination of two sodium-channel blockers can increase neurotoxic adverse events, such as dizziness, diplopia, or ataxia. Therefore, understanding the pharmacodynamics of AEDs is important to design effective and safe combinations of AEDs. New AEDs have unique and multiple mechanisms of actions, which can allow the development of various effective and rational combinations.
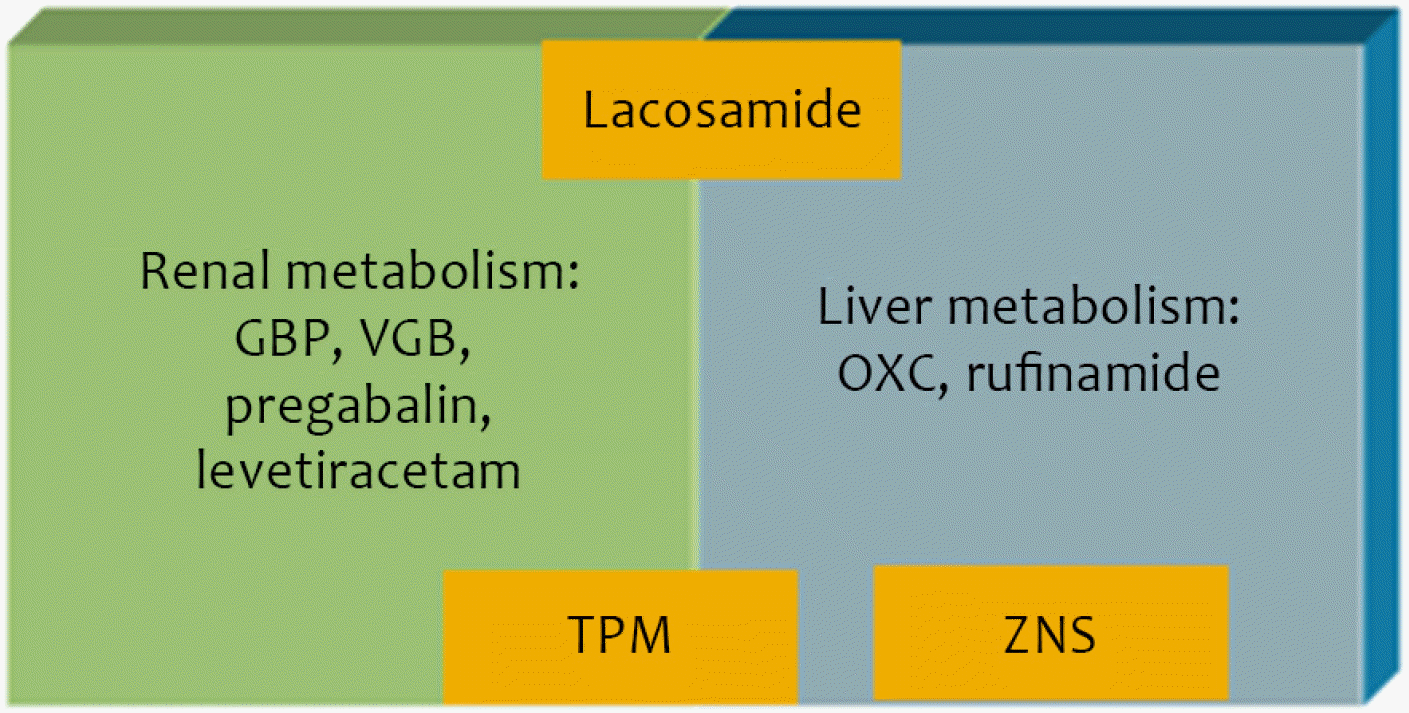
Pharmacokinetic interactions represent the mutual effect of two combined drugs on the absorption, distribution, metabolism, and excretion of drugs. When combining AEDs, the absence of pharmacokinetic drug interactions is easier to handle.14 Several new AEDs do not involve cytochrome p-450 enzyme systems, or do so to a lesser extent (Fig. 4). Other new AEDs are excreted or metabolized through the kidneys. These drugs definitely provide advantages for drug combination. Furthermore, in patients who are taking other drugs to treat comorbid diseases, the absence of drug interactions is important.
Adverse events and outcome measures
As mentioned above, a 50% responder rate is usually used to assess the efficacy of a test drug for the approval of new AEDs. However, the goal of epilepsy treatment is to abolish seizure with minimization of adverse events. A 50% responder rate cannot fully reflect this purpose. There are various outcome measures for AEDs, including percent seizure reduction, responder rate, time to first seizure, presence of adverse events, retention rate, and assessment of quality of life (QOL).5 The treatment goal should be relevant to real-world settings and should include comprehensive measures of efficacy, tolerability, and QOL based on reliable and valid assessment tools. Clinical effectiveness encompasses all these aspects, and the retention rate is very similar to this concept (Fig. 1).
Efficacy is only one aspect that is used to measure the effectiveness of AEDs. For example, the QOL of patients is dependent on seizure freedom, adverse events of AEDs, and presence of anxiety or depression.5,15 QOL improvement occurs primarily among patients who achieve complete seizure freedom. There is no measurable impact of the degree of reduction in seizure frequency on QOL. Adverse events of AEDs and the presence of anxiety/depression are other important factors that affect QOL. Therefore, even though new AEDs cannot achieve seizure freedom, if they have fewer adverse events and play a role in reducing anxiety or depression, they would contribute to the improvement of the QOL of patients.
Individual variability in the response to AEDs
Although the efficacy of new AEDS does not exceed that of old AEDs, on an individual basis, there should be differences in efficacy and in the appearance of adverse events among different drugs. For example, if the efficacy of drugs A and B is similar and one patient does not respond to drug A, this does not necessarily mean that the patient does not respond to drug B (Fig. 5). Therefore, if many drugs are available, the number of therapeutic tools is increased and there is a higher chance of treating patients successfully. Furthermore, the development of a new drug enables a greater number of drug combinations.
Broad-spectrum AEDs are needed for the treatment of generalized epilepsy. Although the effect of valproate is excellent on generalized epilepsies, its use is limited in many patients because of its side effects, such as increased incidence of congenital malformations, low IQ of offspring, and polycystic ovaries.16,17 In that sense, the development of new AEDs with broad-spectrum efficacy is also important.
Women and other issues
The increased incidence of congenital malformations after AED treatment is a serious issue for the treatment of childbearing women. Several nationwide pregnancy registries have been developed to clarify this issue.18–21 To date, some new AEDs have been proven to be relatively safe during pregnancy.
The long-term effects of old AEDs are another issue with this type of drug. Old AEDs, especially those with enzyme-inducing effects, have a negative impact on bone metabolism.22 The use of old AEDs is associated with reduced bone density and increased risk of fracture. New AEDs without enzyme-inducing effects would be relatively safe in this context.
Long-term treatment with old AEDs has been associated with metabolic changes that lead to an increased risk of atherosclerosis in patients with epilepsy.23 Patients who were receiving long-term monotherapy with carbamazepine, phenytoin, or valproate exhibited altered circulatory markers of vascular risk, which was significantly associated with the duration of epilepsy. The effect of new AEDs should be evaluated regarding this matter. It was documented that treatment with new AEDs, such as levetiracetam, oxcarbazepine, and top-iramate, increased the level of low-density lipoprotein, homocysteine, and apolipoprotein B.24
Patients with epilepsy, especially elderly ones, exhibit a high frequency of concomitant diseases, such as vascular, cardiac, and cognitive disorders. These patients usually take various drugs to treat the concomitant diseases. Even healthy elderly subjects are likely to take many drugs. New AEDs without drug interactions are suitable for the treatment of patients with epilepsy who have concomitant diseases.
Conclusions
Even though the efficacy of new AEDs is not stronger than that of old AEDs, there are advantages in using new AEDs and essential needs for their development. There are still many patients who are refractory to established AEDs. When combining AEDs to treat these patients, new AEDs have advantages. They have unique or different mechanisms of action that enable the design of possible synergistic combinations. They usually exhibit fewer or no pharmacokinetic drug interactions. Furthermore, the response to AEDs varies individually. An absence of response to a certain AED does not necessarily mean an absence of response to other AEDs. If many new AEDs are developed, the number of therapeutic weapons available to obtain this individual response is increased. The appearance of adverse events of AEDs strongly affects the QOL of patients. Many new AEDs with fewer adverse events, including the induction of congenital malformations, have been developed. Other concerns about the long-term effect of established AEDs, such as bone health and the development of atherosclerosis, may be alleviated by the use of new AEDs.