Introduction
Epileptic seizures, in general, are not an uncommon event, with an overall annual incidence of approximately 30/100,000 for newly diagnosed epilepsy and 55/100,000 for unprovoked seizures, respectively.1 In most instances, normal physiological and biochemical mechanisms, such as the loss of ionic gradients, depletion of energy substrates and glutamate, as well as glial buffering and changes in local neuromodulators cause seizures to be self-limiting, frequently not necessitating emergency pharmaceutical therapy.2
In contrast, when generalized convulsive seizures do not spontaneously terminate after 5 minutes or the patient does not regain consciousness between recurrent seizures, the term status epilepticus (SE) is used.3 The annual incidence of SE is reported to be 8.5–41/100,000, and its mortality ranks high at approximately 10–33%.4,5 By definition, SE requires an immediate and aggressive pharmaceutical intervention to prevent permanent neural damage and organ failure, and general prognosis is known to deteriorate rapidly with ongoing seizure activity. Current guidelines on SE suggest the use of benzodiazepines as a first-line therapy. If this fails in terminating the seizure, antiepileptic drugs (AED) such as fosphenytoin, valproic acid, phenobarbital, levetiracetam, or lacosamide are then to be given intravenously.6 If these first- and second-line anticonvulsant therapies are exhausted and epileptic activity continues (which is the case in approximately 23% of SE7), the criteria for refractory status epilepticus (RSE) are then met, and evidence-based therapeutic strategies become sparse. General anesthesia with midazolam, propofol, or thiopental is widely recommended while continuous infusion of AEDs is maintained.6,8 Burst-suppression-pattern (BSP) anesthesia should be achieved and maintained for at least 24 hours while adequate serum levels of anticonvulsant agents are further established. After this, anesthesia is then slowly reduced under close electroencephalographic (EEG) monitoring. Further pharmacological treatment options in RSE include the continuous infusion of the N-methyl-D-aspartate (NMDA)-antagonist ketamine or titration of inhaled anesthetics such as isoflurane to induce a BSP on EEG.9 For instances in which, despite these extensive efforts, RSE either continues or recurs for a more than a 24-hour period, the term super-refractory status epilepticus (SRSE) was coined.8
The etiology of (S)RSE differs from that of other seizures in that there is often no preexisting epilepsy, and acute events such as trauma, stroke, infectious disease, metabolic causes, and sepsis present more frequently.10 In addition, refractory seizures can affect previously healthy patients without an obvious cause, apparently sometimes due to cryptogenic autoimmune diseases.11 While symptomatic treatment of SE is, of course, urgent, identification of the underlying cause and targeted therapy are of great importance and crucial to the overall outcome.12
Prolonged seizures as in SE have a tendency to become both self-sustaining and more resistant to pharmacotherapy, which may in part be due to the preponderance of the more severe etiological factors seen in them.13 Pharmaco-resistance in longer lasting seizures is found especially for benzodiazepines, barbiturates, and propofol, which is attributed to a gradual downregulation in functional Gamma-Aminobutyric acid (GABAA)-receptors over the course of the seizure.14,15 GABA is known as the principal inhibitory neurotransmitter in the central nervous system. Other factors that may contribute to seizures becoming self-sustaining include excitotoxicity through up-regulation of NMDA receptors and excessive glutamatergic stimulation, promoting neural damage by intracellular calcium influx, inflammatory reactions, emerging dysfunction of the blood-brain-barrier, maladaptive changes in neuropeptide modulators, and – over time – altered gene expression.12,13,16
Due to these self-sustaining properties and emerging pharma-co-resistance, therapeutic strategies other than anticonvulsant agents merit further attention in (S)RSE. Among those are treatments such as hypothermia (which is supposed to achieve a reduction of cerebral metabolism), ketogenic diet (in which an anti-inflammatory mode of action is suspected), as well as steroids and immunotherapies (even in the absence of a detectable autoimmune disease).12 However, evidence for all these strategies rests only on a limited number of case studies.
Electroconvulsive therapy (ECT) is known as a further possible therapeutic tool for (S)RSE and has been successfully applied in a number of such cases (for a current review see17). Evidence for the anticonvulsant properties of ECT reaches back as early as the 1930s18 and has often been replicated by finding a gradual increase in the seizure threshold over the routine course of ECT for psychiatric indications.19–22 Such an increase in seizure threshold has been found in a majority of patients, exceeding 94% in a recent study, but being highly variable in magnitude.19 The exact mechanism of the proposed anticonvulsant properties of ECT remains unclear to date. Possible modes of action include the activation of endogenous GABAergic pathways, promotion of neurotrophic factors and their connected effects on synaptic neuroplasticity, as well as a more general decrease of neural metabolism.23,24
In comparison to ECT, other modes of noninvasive brain stimulation, such as transcranial magnetic stimulation9 or transcranial direct current stimulation,25 while certainly representing promising techniques for the treatment of epileptic disorders in the future, are as yet far less established in the treatment of RSE.
Despite some encouraging results with previous utilization of ECT in the indication of (S)RSE, the ideal stimulation parameters, e.g., electrode position or stimulation energy as well as treatment schedules, are still under debate. Currently, controlled clinical studies investigating these important issues are still lacking. Some evidence points to a more favorable outcome when a more intensified ECT protocol is applied. For example, in a recent case study by Kamel et al.,26 the induction of seizures three times per day on several consecutive days in (S)RSE was found to be effective in two of three cases.
Case
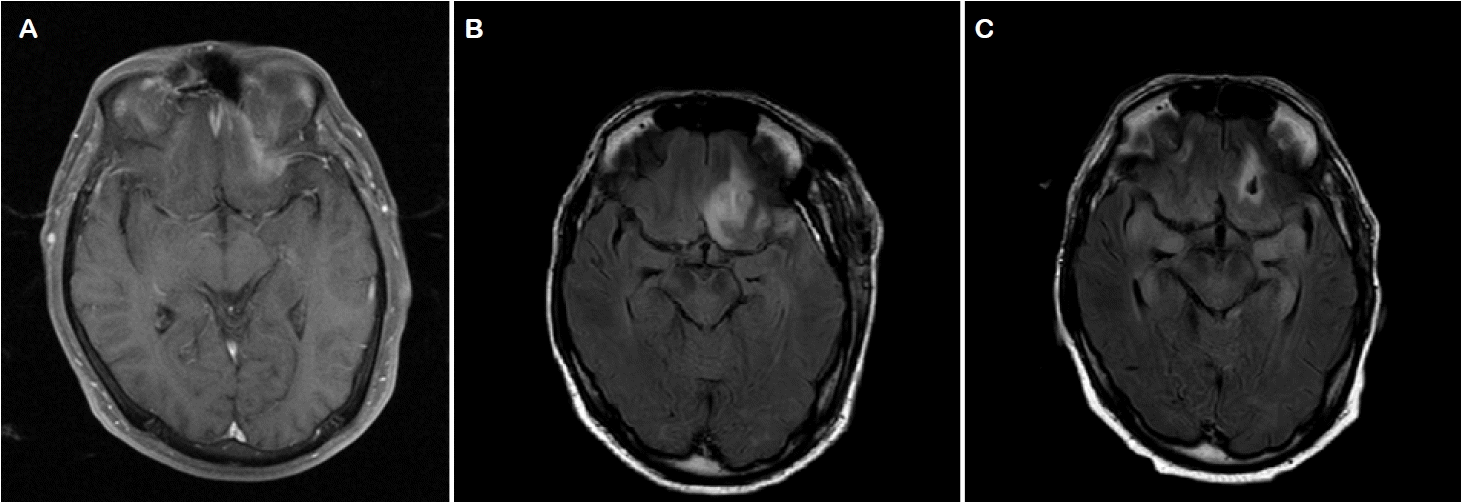
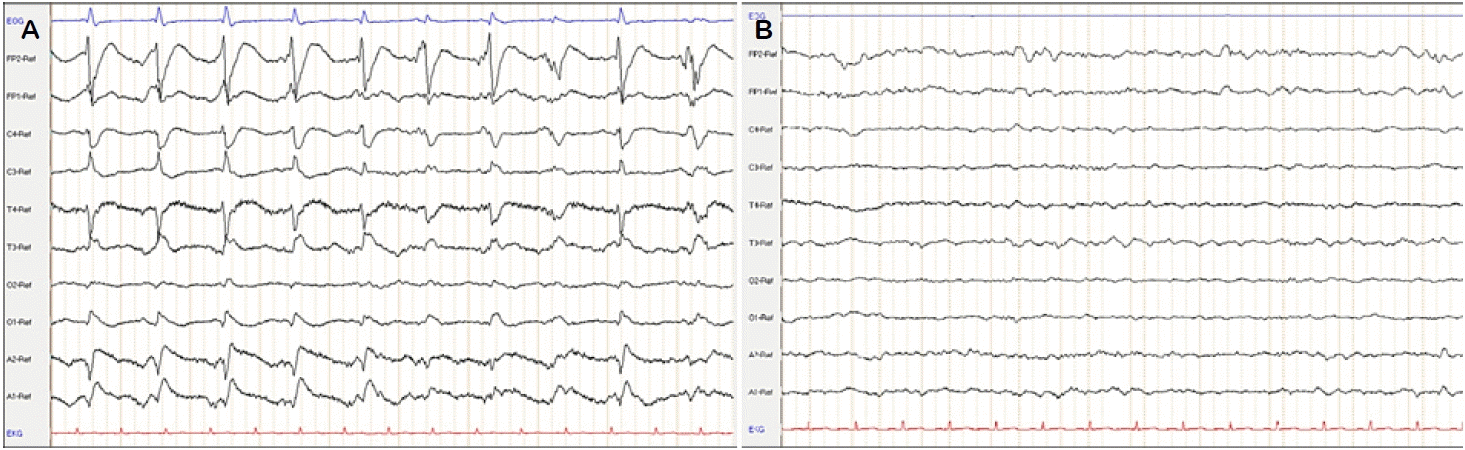
We report on a 61-year-old male patient who was diagnosed with an adenocarcinoma originating from the paranasal sinuses that had been infiltrating the base of the skull and compressed the N. opticus bilaterally (Fig. 1A), giving rise to impaired vision and headaches. After the malignant tumor had been removed microsurgically and subsequent radiochemotherapy had been administered (Fig. 1B), the patient was admitted to the hospital approximately three months later due to confusion and impaired consciousness, which declined rapidly into a comatose state. The EEG revealed a non-convulsive SE with continuous (poly)spike-wave-complexes bilaterally but most pronounced right-frontally (Fig. 2A). Initial treatment comprised the administration of levetiracetam, lacosamide, and phenytoin intravenously. PCR from the cerebrospinal fluid (CSF) was positive for herpes simplex virus (HSV) type 1 DNA, and magnetic resonance imaging showed localized hyperintensities in the temporal and temporopolar regions (Fig. 1C). Based thereon, HSV encephalitis was suspected as the underlying diagnosis despite normal CSF white cell count and protein-levels, and the patient was started on an intravenous course of aciclovir. Epileptic activity persisted despite establishing an extensive AED regimen, including midazolam, lacosamide, levetiracetam, ketamine, valproate, zonisamide, and phenytoin (with multiple substances used simultaneously at any time; Fig. 3). Burst suppression anesthesia was induced multiple times using either midazolam/ketamine, thiopental, or isoflurane for at least 48 hours while high anticonvulsant medication serum levels were sustained. Yet, attempts to wean the patient off the anesthesia failed repeatedly because of recurring SE, which resulted in the need for ongoing anesthesia and tracheotomy. Remission of epileptic activity could only be achieved temporarily for up to 5 days at most, with severely abnormal EEG-findings persisting during these times. In addition, two courses of high dose cortisol for suspected autoimmune encephalitis, despite negative anti-neural antibody panels, as well as high dose magnesium failed to induce significant improvement. After 44 days of persisting SRSE, the commencement of ECT was decided.
Methods
As preparatory measures prior to ECT, the anticonvulsant treatment regimen was deescalated, with all anticonvulsant agents except for levetiracetam and ketamine being discontinued (Fig. 3). Written informed consent for the procedure was obtained from the patient’s next of kin. During the first course of ECT, no anesthesia was applied due to the comatose state of the patient. During the second course of ECT, propofol (100–150 mg) was used as a narcotic since the patient opened his eyes but still remained fully unresponsive. We chose rocuronium (100 mg) as a relaxant instead of succinylcholine because of the patient’s prolonged immobility in order to minimize the risk of exceeding hyperkalemia. Muscular relaxation was controlled via repeated Train-Of-Four (TOF) measurements. We did not use electromyographic (EMG) monitoring since an extensive cuffing period would have been required due to the repeated seizure inductions during a single ECT session. However, a mild generalized convulsive activity could be observed throughout the treatment sessions since muscle relaxation was not fully complete. Using controlled hyperventilation during mechanical ventilation (BiPAP), the pCO2 was kept under 30 mmHg. On each of 4 consecutive days, 3 generalized seizures were induced, with stimulation intervals of 3–4 minutes, following the treatment protocol introduced by Kamel et al.26 The Thymatron-IV® system (Somatic, LLC, Lake Bluff, IL, USA) was used on a double-dosage setting at the maximum 200% stimulation intensity (equivalent to a mean charge of 1,031 mC). Standard bitemporal electrode placement was used. The applied current was held constant at 920 mA, with a pulse frequency of 70 Hz and a pulse width of 1 msec. Seizure activity was detected by constant two-lead EEG monitoring as provided by the Thymatron-IV® system (Somatic, LLC).
Results
The overall seizure quality was moderate in terms of amplitude (mean maximum amplitude, 245 μV; standard deviation [SD], 129 μV) and ictal bihemispheric coherence. Vegetative reactions marked by a significant increase in heart rate as well as a postictal suppression were observable in most of the induced seizures (59% and 79%, respectively). The duration of seizures varied from 16 to 52 seconds (mean duration, 26.7 seconds; SD, 8.1 seconds). There was no difference in seizure quality parameters between the two courses of ECT treatments. By way of example, a representative treatment session as recorded by the ECT device is depicted in Fig. 4. During the first course of ECT, seizure cessation was achieved after the fourth day of treatment; the patient opened his eyes spontaneously and was able to fixate his gaze. However, epileptic activity and a comatose state recurred five days after ending ECT despite reconstitution of the AED treatment comprising levetiracetam, lacosamide, phenobarbital, zonisamide, and lorazepam. Continuous infusion of midazolam/ketamine with burst suppression was restarted. A second course of ECT was administered after another ten days. At this time, the induction of the identical ECT protocol on each of the 4 consecutive days was followed by three tapering sessions every other day. A stable remission of seizure activity was achieved after the third tapering treatment session (Fig. 2B), 10 days after reintroduction of ECT. The patient was awake and able to perform purposeful movements and utter some adequate verbal expressions. Shortly thereafter he was fit to be transferred to a rehabilitative facility. The patient remained in remission from recurrent SE as assessed by follow-up several months later, although considerable impairment in communicative abilities remained.
Discussion
Until now, there have been no randomized controlled trials examining the efficacy of ECT in the treatment of (S)RSE. In some recent reviews, a number of retrospective case reports have been published and summarized.17,27,28 The ECT protocols used varied greatly in terms of the total numbers of sessions as well as the number of seizure inductions per session. Furthermore, differential effects of the stimulation location, the charge applied, the timing of ECT in relation to the duration of (S)RSE, as well as the concomitant use of anticonvulsant medication are largely unknown. In addition, the interpretation of the effect of ECT is further complicated by the heterogeneous etiologies in the cases reported and the application of multiple disease-specific therapies at the same time.
Lambrecq et al.27 in their review of eleven cases of ECT in RSE concluded a rather favorable outcome, with ECT being successful in the termination of SE in 80% of cases and full functional recovery being achieved in at least three of them (27%). Clinical improvement was generally achieved during the first 10 days of treatment. However, the reporting bias associated with case reports or series is known to overestimate the potential beneficial effect, and thus these results should be interpreted with caution.
A more recent review by Zeiler et al.17 included fourteen studies reporting about nineteen patients. Seizure reduction or control was achieved in 58% of cases. Relative safety of the procedure was stated, with few adverse events reported, such as lethargy or amnesia. Yet, 10 of 13 patients for whom outcome was reported fell into the category of dead or severely disabled. The authors concluded that, at present, only limited evidence exists for the effectiveness of ECT in the treatment of (S)RSE. Although a potential impact of ECT on seizure control was seen, the routine use of ECT would not be recommended in light of the available data.
In deciding on the most promising stimulation parameters to terminate SRSE in the patient described, we especially took into consideration the study of Kamel et al.,26 in which stimulation parameters were well described, and an overall favorable outcome was described in two of the three cases. The course of treatment included daily sessions of ECT performed on consecutive days with each session comprising at least three seizure inductions. The mean charges applied of approximately 507 mC corresponded to the standard maximum output of the Thymatron-IV® system (Somatic, LLC) available in the USA and Canada (100% stimulation energy).
Only recently, Ahmed et al.28 conducted the largest case series on the effects of ECT in SRSE to date. This study showed improvement in five of eight cases in SRSE of various etiologies using bitemporal stimulations with a mean stimulus intensity of 504 mC and mostly two to three stimulations per daily session for at least three consecutive days. However, seizure inductions were not completely reliable, as evidenced by a mean of 4.5 seizures per case induced by a mean of 9 stimulations per case. As was done in our study, the anticonvulsant medication was continued in most cases.
Considering that the reliable induction of seizures is of utmost importance for the efficacy of the ECT treatment in SRSE, we favored stimulation charges at intensities much higher than those applied by Kamel et al.26 and Ahmed et al.28 Since a “double dose stimulus program” is approved for use with the Thymatron-IV® system (Somatic, LLC) in Europe (200% stimulation energy), we were able to apply a mean output charge of 1,031 mC. Using this stimulus setting, we were able to achieve a highly reliable induction of seizures associated with the repeated induction of a postictal suppression despite the continuation of a potent anticonvulsant medication regimen.
Of note, stimulation parameters used here differed considerably from those used in the treatment of major depressive disorders via ECT. The induction of a seizure with adequate duration and overall quality is usually aimed for at the least required stimulation energy in order to minimize dose dependent side effects. Such side effects include neurocognitive impairments, especially with regards to episodic memory, and are usually of transient nature.29 We considered those dose dependent and transient side effects to be negligible in the given case with respect to the life-threatening condition of the patient.
As noted by Cline and Roos30 in their case report, the poor functional outcomes seen in many patients may be due to the rather late use of ECT in the course of SE. Zeiler et al.17 found the interval between SE onset and initiation of ECT to be 44 days on average in the cases reviewed. After such time, reduced functional outcomes may largely be caused by the prolonged seizure itself, as evidenced by findings of progressive brain atrophy in the course of ongoing SE.16,31 Hence, the best point in time for the escalation of the anticonvulsant efforts towards ECT might be crucial, yet still undetermined.
Due to the heterogeneity of ECT parameters in the reports available, and as previous reports often lacked a thorough description of the parameters used, no general recommendations for the exact mode of application can be given with certainty. This holds true especially for the localization of the stimulus, its intensity, and its frequency. On a similar note, there are no clinical data available to support or discourage the use of ECT in a particular group of patients with (S)RSE, e.g., such as with or without structural cerebral lesions, and no recommendation can be made concerning the maintenance or discontinuation of the concomitant anticonvulsant medication.
In the case reported here, the etiology of the SRSE could not be identified with certainty. In addition to the suspected HSV encephalitis (in which a second PCR from CSF was negative; however, and CSF cell count remained in the normal range), a structural cause (i.e., the tumor and/or the defect due to surgery), as well as limbic encephalitis (despite the fact that specific antibodies could not be detected and the immunosuppressive treatment approach remained ineffective) need to be taken into consideration. It has to be conceded that a consolidation of the affected structural lesion over time could be of substantial importance for the clinical improvement. Nonetheless, the repeated temporal correlation of the initiation of the ECT course and the clinical response remains remarkable.
On a theoretical level, it can be speculated that for the anticonvulsant properties of ECT to emerge, the repeated induction of postictal suppression (as observable via EEG) is the key factor, which brings forth the activation of endogenous GABAergic pathways and the concomitant gradual increase of seizure threshold over time, as seen in patients receiving ECT for psychiatric illness. The relationship between the antidepressant and anticonvulsant properties of ECT remains unclear until now. Both seem to be brought on over time, necessitating multiple treatments for the respective indications. While Sackeim et al.20 found a correlation between the increase of seizure threshold and the improvement of mood in depressed patients and thus postulated the emergence of central inhibitory neurotransmission processes to be crucial to both properties, the majority of other studies (while strongly supporting the finding of an increase in seizure threshold over time) did not find a clear relationship between anticonvulsant and antidepressant effects.19,21,32 Duthie et al.19 suspected that, while the anticonvulsant properties of ECT were not the decisive factor for its antidepressant efficacy, they might represent a marker for neurophysiological changes taking place, which might be crucial for both anticonvulsant and antidepressant effects. In particular, the increase in brain-derived neurotrophic factor, which is associated with ECT, might be involved in both its anticonvulsant and antidepressant properties, as it promotes neurogenesis as well as synaptic plasticity.33 Since a gradual up-regulation of GABAA-receptors is hypothesized to occur over the course of ECT, this might also diminish the pharmaco-resistance at least for certain AED regimes, e.g. benzodiazepines, barbiturates, and propofol.14,15
With our report, we provide further evidence that (S)RSE cessation can successfully be achieved by means of high-intensity ECT, even after six weeks of prolonged SE and exhausted anticonvulsant pharmacotherapy. The intensified ECT regimen applied here, including daily treatment sessions at high energy charges with repetitive seizure stimulations in each session may have contributed to the favorable outcome. The relative significance of a high-intensity ECT protocol as well as the role of ECT in relation to other advanced treatment options for (S)RSE (such as ketogenic diet or hypothermia), however, have yet to be determined.